Skyhawk Therapeutics Announces Nine Month Interim Results in Patients from its Phase 1 Clinical Trial of SKY-0515 as a Treatment for Huntington's Disease
27.01.2026 - 14:05:22 | dgap.de| Skyhawk Therapeutics / Key word(s): Miscellaneous 27.01.2026 / 14:05 CET/CEST The issuer is solely responsible for the content of this announcement. Nine-month findings show mean improvement in Composite Unified Huntington's Disease Rating Scale from baseline of +0.64 points, compared to natural history expected worsening of cUHDRS in symptomatic patients of -0.73 points over nine months, based on propensity score weighting.Skyhawk also announces SKY-0515's Phase 2/3 FALCON-HD trial has expanded worldwide. Skyhawk has now dosed more than 90 patients.BOSTON, Jan. 27, 2026 /PRNewswire/ -- Skyhawk Therapeutics, Inc., a clinical-stage biotechnology company developing novel small molecule therapies to modulate critical RNA targets, today announces positive results from the nine month interim analysis of the Company's investigational treatment for Huntington's disease (HD) with SKY-0515. 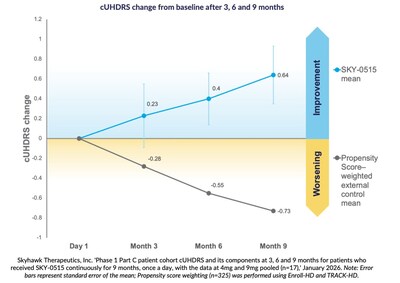 SKY-0515's Phase 1 clinical trial is a first-in-human trial designed to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of SKY-0515 in healthy volunteers and individuals with early-stage Huntington's disease (HD). The trial is separated into three parts. Parts A and B evaluated SKY-0515 in Healthy Volunteers. Part C is a double-blind placebo-controlled parallel design study of two dose levels of SKY-0515 and placebo in individuals with early-stage HD (HD-ISS Stage 1, 2, or mild Stage 3) for 84 days followed by a 12 month extension of active treatment where all participants will receive either a low or high dose of SKY-0515 in a blinded fashion. The objectives of the study include evaluating mutant HTT protein and PMS1 mRNA. The first patients were dosed in SKY-0515's Part C in January 2025. Enrollment in Phase 1C of the SKY-0515 trial is now complete.About SKY-0515's Phase 2/3 FALCON-HD Clinical Study FALCON-HD (NCT06873334) is a Phase 2/3 randomized, double-blind, placebo-controlled, dose ranging study to evaluate the pharmacodynamics, safety, and efficacy of SKY-0515 in 120 participants with Stage 2 and early Stage 3 HD across 12 sites in Australia and New Zealand, and 400 participants with Stage 2 and early Stage 3 HD in 40+ worldwide sites. Eligible patients will receive a once-daily oral dose of SKY-0515 at one of three dose levels or placebo, for a treatment period of at least 12 months. The trial aims to assess the potential of SKY-0515 to modulate RNA splicing and reduce mHTT and PMS1 proteins, which are implicated in the pathology of Huntington's disease. Additional information about FALCON-HD, including participating sites and eligibility criteria, can be found at ClinicalTrials.gov and www.FALCON-HD.com.About Skyhawk Therapeutics Skyhawk Therapeutics is a clinical-stage biotechnology company which uses its proprietary platform, SKYSTAR®, to discover and develop small molecule RNA modulating therapies for the world's most intractable diseases. For more information visit www.skyhawktx.com.Skyhawk Contact Maura McCarthy Head of Corporate Development maura@skyhawktx.com  Logo - https://mma.prnewswire.com/media/710814/Skyhawk_Therapeutics_Logo.jpg  27.01.2026 CET/CEST Dissemination of a Corporate News, transmitted by EQS News - a service of EQS Group. The issuer is solely responsible for the content of this announcement. The EQS Distribution Services include Regulatory Announcements, Financial/Corporate News and Press Releases. View original content: EQS News |
So schätzen die Börsenprofis Aktien ein!

Seit 2005 liefert der Börsenbrief trading-notes verlässliche Anlage-Empfehlungen – dreimal pro Woche, direkt ins Postfach. 100% kostenlos. 100% Expertenwissen. Trage einfach deine E-Mail Adresse ein und verpasse ab heute keine Top-Chance mehr. Jetzt abonnieren.
Für. Immer. Kostenlos.
Für. Immer. Kostenlos.
boerse | 68524621 |

